Human Cerebral Cortex Organization Characterized by Functional PET-FDG “Metabolic Connectivity”
Abstract
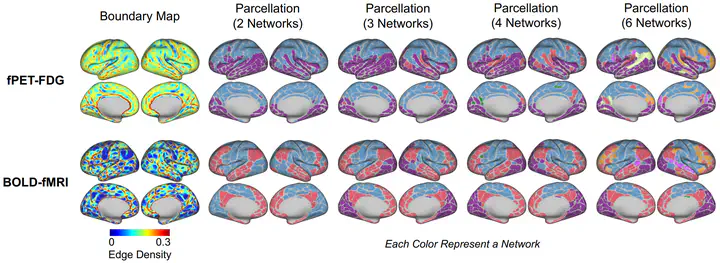
Purpose: In this study, we characterize the spatiotemporal organization of resting-state metabolic connectivity (RSMC) in the human brain, as measured by [18F]- fluorodeoxyglucose (FDG) functional PET (fPET-FDG). We examine the relationship between RSMC organization and resting-state functional connectivity (RSFC) derived from functional magnetic resonance imaging and other known cortical organizational principles. Methods: Resting-state fPET-FDG data from 24 individuals were obtained from a publicly available repository. We characterized local metabolic organization using connectivity-based boundary mapping, with adaptations to account for the low signal-to-noise ratio of fPET-FDG data. We then estimated global metabolic organization through community detection-based network and principal gradient analyses. Furthermore, we examined how metabolic connectivity is shaped by temporal-frequency-specific components of fPET-FDG signal. Finally, we contextualized metabolic organization by relating metabolic gradients to anatomical, functional, and energetic reference measures. Results: At the local scale, boundary mapping results indicated structured transitions shaped by a combination of both fast and slow fPET-FDG signals, partly overlapping with RSFC boundary maps. Globally, RSMC analyses revealed a robust metabolic structure organized along a superior-inferior cortical gradient. This pattern remained consistent across network community detection and principal gradient analyses and was primarily driven by low-frequency, minute-scale fPET-FDG dynamics. The identified large-scale metabolic profile aligns closely with several known anatomical and energetic constraints. Conclusion: This study characterizes the spatiotemporal organizational principles of RSMC, deepening insight into the brain’s energetic framework and providing a basis for future cognitive and clinical investigations of metabolic connectivity organization. https://www.biorxiv.org/content/10.64898/2026.02.15.706044v1